Frauenhofer Publica / MCC
LEVERAGING PATIENT DATA REPOSITORIES TO ADVANCE MEDICAL CANNABINOID RESEARCH
Research poster, which stands at the core of Copeia/pep's mission and vision, presented at the 5th Medicinal Cannabis Congress (MCC 2024), held in Berlin on May 23-24, 2024, indexed in the Fraunhofer Publica repository
A.Landschaft (1,2), A.Ihlenfeld (2), G.Hirt (2)
Fraunhofer-Institut für Intelligente Analyse- und Informationssysteme (IAIS), Sankt Augustin, Germany
Copeia GmbH, Bergisch Gladbach, Germany D-51469
Methods:
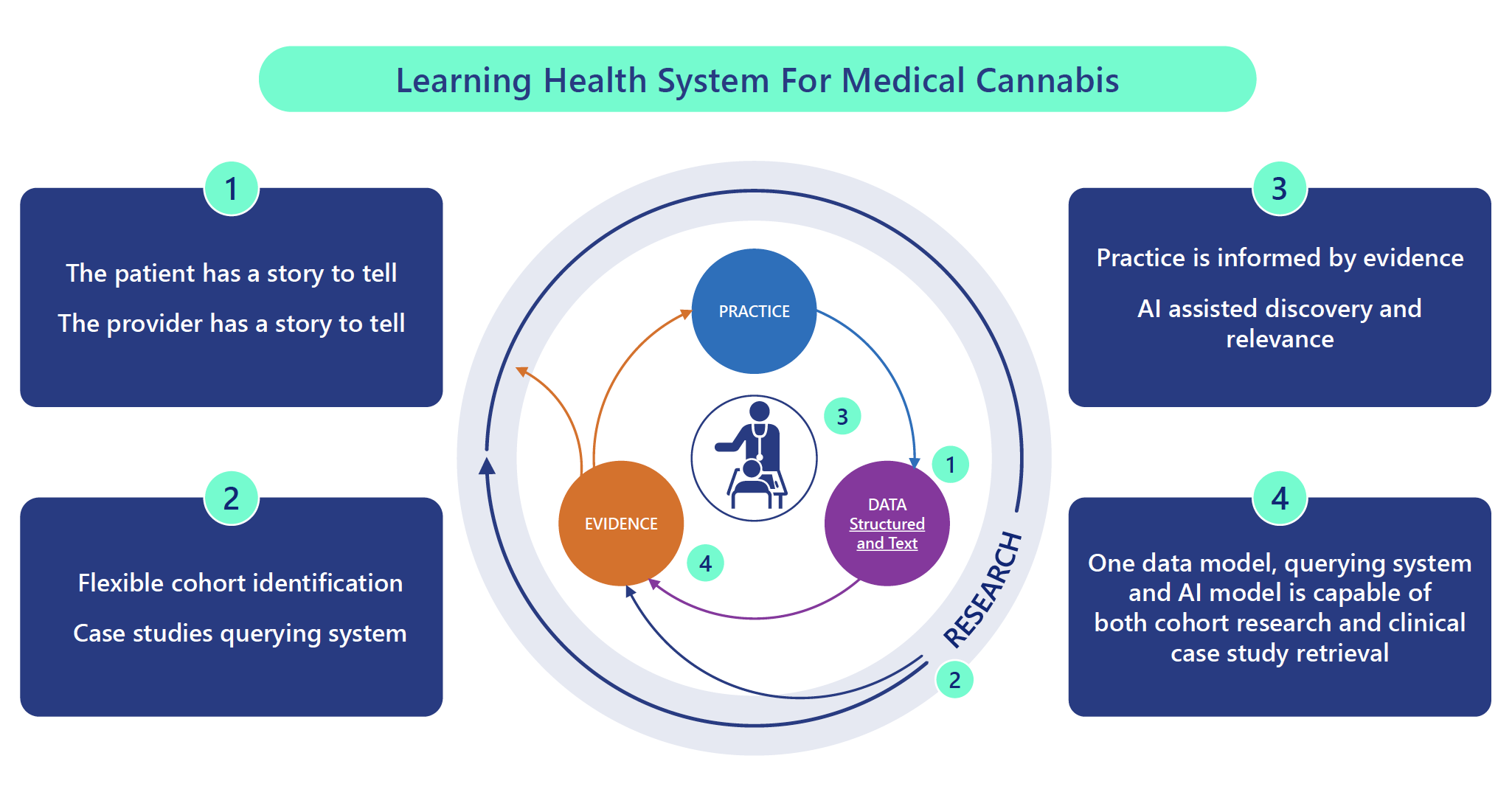
Databases and electronic journal collections were searched for articles, out of which we extracted 34 highly relevant studies to Real World Evidence (RWE) Repository for medical cannabis. We divided the articles into 4 groups: 1. Real World Evidence in Medical Cannabis Research (paucity of studies and Benefits of RWE) 2. Healthcare Repositories That Eliminated Research Bottlenecks and Strategies That May Apply to Cannabis Clinical and Research Challenges 3. Usage of ML to Derive Clinical Insights for Cannabinoid Therapies 4. Importance of Case Studies and Anecdotes for Emerging Interventions Where Strong Evidence Is Lacking. For each relevant group, we extracted functional and research features relevant to leveraging patient data repositories to advance medical cannabinoid research. Our methodology comprised these four key components/ groups. Firstly, we focused on the benefits of RWE, along with the quantity and quality of RWE studies linked with medical cannabis research. Secondly, we identified primary gaps in cannabinoid and medical cannabis research, emphasizing the lack of robust evidence and patient data. Thirdly, we conducted a comprehensive review of existing healthcare repositories, drawing parallels between their objectives, methodologies, and impact. Finally, we explored the potential of leveraging artificial intelligence (AI) alongside these repositories to enhance data analysis and interpretation.
Results:
Despite the increasing interest in medical cannabis, the lack of real-world data from randomized controlled trials poses challenges in assessing its efficacy. Recent RWE studies offer a promising alternative to traditional trials, aiming to fill crucial gaps in understanding medical cannabis's therapeutic benefits and safety profiles (Banerjee et al., 2022; Rapin et al., 2021; Shakeri et al., 2021; Sakal et al., 2022; Schlag et al. 2022). Through our analysis, we emphasize the critical need for repositories that provide researchers and clinicians access to real-world patient data that encompasses the entire spectrum of cannabis usage in clinical settings. We identified 16 repositories that effectively addressed research obstacles, similar to those encountered in cannabis research, by incorporating real-world datasets (See Table 2). In various medical domains, parallels can be drawn to the utilization of data repositories containing RWE to effectively address analogous challenges encountered within the realm of medical cannabis research (e.g. PECARN, SEER, MIMIC-III, INSIGHT-GHT, etc.). More specifically, INSIGHT-GHT is an ongoing initiative that comprehensively captures all clinically relevant aspects of administering somatropin to treat growth hormone (GH) deficiency. It informs researchers and clinicians about interventions that enhance disease outcomes and ranks new GH products. We also observed how AI-driven algorithms are facilitating pattern recognition, predictive modelling, and personalized cannabinoid-based treatment recommendations for conditions such as autism and Parkinson’s disease leveraging patient data repositories (Quillet et al., 2023; Yucel, Ozcelik, & Algul, 2023). Furthermore, drawing from various studies (Crowe et al., 2011; Enkin & Jadad, 1998; Jackevicius, 2018; Nissen & Wynn, 2014; Paparini et al., 2020), it is indicated that case study research offers valuable insights complementing the limitations of randomized controlled trials, contributing to a deeper understanding of complex issues in real-life settings and playing a crucial role in clinical practice.
Conclusions:
Our study underscores the critical significance and feasibility of establishing a dedicated repository of real-world patient data to drive innovation in and advancement of medical cannabinoid research. To address this challenge, we propose the development of a case study repository for medical cannabis, combined with AI analytic and search-engine tools. This integration would enable clinicians and scientists to efficiently locate relevant case studies to complement clinical data and evidence-based treatment guidelines. This data, combined with ML analytic tools, can accelerate medical cannabis research by predicting the effectiveness of cannabinoid compounds, and discovering new therapeutic applications for cannabinoids. Moreover, leveraging AI and large RWE to efficiently search relevant case studies will allow researchers to ascend the Hierarchy of evidence and unlock the full potential of cannabinoid therapy and improve patient outcomes. A comprehensive clinical data repository, along with a case-study search engine, could further facilitate the development of effective cannabinoid therapies. This would be achieved by providing RWE data in instances where controlled clinical studies are deficient, and by supplementing clinical trial results and evidence-based care guidelines with contextual and transferability data. The ability to efficiently search a repository of case studies will also help researchers identify novelties, limitations of clinical trials, and potential cannabinoid mechanisms and applications. In scenarios characterized by the absence of evidence-based clinical protocols, individual case studies prove valuable in both clinical practice and research environments. By leveraging AI, researchers can unlock the full potential of cannabinoid therapy, ultimately leading to evidence-based practices and improved patient outcomes. AI and ML technologies aid in ascending the hierarchy of evidence, transitioning from individual case studies to the establishment of evidence-based clinical protocols.
Keywords:
cannabinoids, medical cannabis research, real-world patient data, data repositories, AI, ML, evidence-based medicine
If you would like to learn more about Copeia, have questions or suggestions, please contact us by email and we will get back to you as soon as possible. We look forward to hearing from you.
Your Copeia team
Copeia’s digital health solutions evolve personalised treatment of cannabis patients
Copeia combines expertise from international cannabinoid research with practical knowledge of doctors, patients, pharmacies and producers.
Copeia is a software company based in Cologne and Berlin that develops AI-based products and workflow solutions to support medical cannabis care.